The Longevity Revolution Is Here — But Not for Everyone
The science of extending human life has advanced more in the past three years than in the prior three decades, yet the emerging longevity revolution threatens to become the defining inequality of the 21st century.
The science of extending human life has advanced more in the past three years than in the prior three decades, yet the emerging longevity revolution threatens to become the defining inequality of the 21st century. Between 2023 and 2025, GLP-1 receptor agonists demonstrated a 20% reduction in major cardiovascular events, the first CRISPR-based gene therapy won FDA approval, and private investment in longevity science surged to $8.49 billion — a 220% increase over 2023. Simultaneously, the life expectancy gap between the richest and poorest Americans widened to nearly 15 years, Medicare's trust fund moved its projected depletion date forward to 2033, and federal research funding faced unprecedented cuts. A generation of young Americans is spending $214 per month on biohacking while 25% of their peers delay basic medical care due to cost. This is the central paradox of our moment: the tools to extend human healthspan are arriving faster than our systems — ethical, economic, and political — can absorb them.
Part I: The Science — Breakthroughs, Blind Spots, and the Distance Between Mice and Men
The Aging Framework Gets an Upgrade
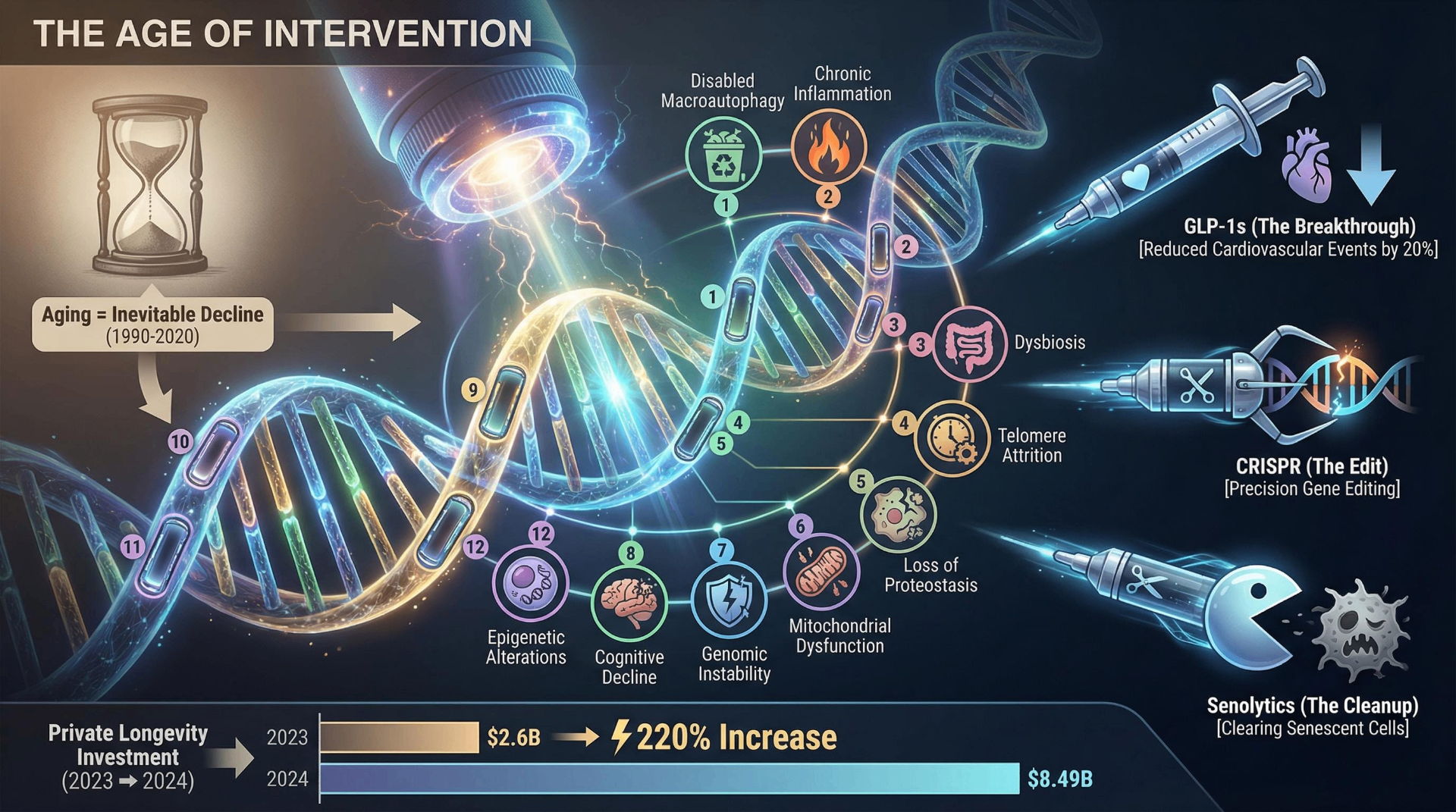
The conceptual foundation of longevity science shifted in January 2023, when Carlos López-Otín, Maria Blasco, Linda Partridge, Manuel Serrano, and Guido Kroemer published "Hallmarks of aging: An expanding universe" in Cell. The paper expanded the original 2013 framework from 9 to 12 hallmarks of aging, adding disabled macroautophagy, chronic inflammation, and dysbiosis (gut microbiome disruption). Cited over 11,000 times, the updated framework reflects a maturing field that increasingly views aging not as inevitable decline but as a constellation of targetable biological processes.
This shift from describing aging to intervening in it has catalyzed work across multiple fronts. Senolytics — drugs that clear senescent "zombie" cells — entered over 30 clinical trials by 2025. The most rigorous, a Phase 2 randomized controlled trial led by Joshua Farr at the Mayo Clinic, tested dasatinib plus quercetin in 60 postmenopausal women. Published in Nature Medicine in 2024, results were sobering: no significant benefit in the overall population. But among women with the highest senescent cell burden, bone formation markers improved by 34% and bone mineral density increased by 2.7%. The lesson, as Khosla, Monroe, and Farr argued in Nature Aging (2025), is that senolytics may require a personalized approach — targeting patients with measurably high senescent cell loads rather than treating aging as monolithic.
Epigenetic clocks, the field's most important measurement tools, continue to sharpen. Daniel Belsky's DunedinPACE (Columbia University), which measures the rate of aging rather than biological age, has become the preferred instrument in longevity research. GrimAge, a second-generation clock, predicts mortality with striking accuracy. Yet controversy persists about how these tools are used — and misused. When Bryan Johnson claimed he had reduced his "pace of aging by 31 years," Morgan Levine, a pioneer in epigenetic clock development, publicly corrected the record: epigenetic clocks measure rates, not years, and his claim "appears unsubstantiated."
GLP-1 Drugs Deliver the Decade's Most Consequential Clinical Results
No class of drugs has reshaped the longevity conversation more dramatically than GLP-1 receptor agonists. The SELECT trial, published in the New England Journal of Medicine in November 2023, enrolled 17,604 patients with established cardiovascular disease and obesity but without diabetes. Randomized to semaglutide 2.4 mg weekly or placebo for a mean of 39.8 months, the semaglutide group experienced a 20% reduction in major adverse cardiovascular events (HR 0.80, 95% CI 0.72–0.90, P<0.001), average weight loss of 9.4%, and a 19% reduction in all-cause mortality (HR 0.81). For patients with heart failure, the benefit was even more pronounced (HR 0.72). These are not marginal gains — SELECT demonstrated that a weight-loss drug could independently reduce heart attacks, strokes, and death.
The SURMOUNT trials of tirzepatide (Eli Lilly's dual GIP/GLP-1 agonist, marketed as Mounjaro and Zepbound) pushed the envelope further. At 72 weeks, participants lost 15–21% of body weight depending on dose. A DXA substudy published in 2025 confirmed that approximately 75% of weight lost was fat mass, partially addressing fears about muscle wasting. A separate MRI substudy in The Lancet Diabetes & Endocrinology showed tirzepatide actually reduced muscle fat infiltration, suggesting "adaptive body remodeling" rather than indiscriminate tissue loss.
Concerns persist, however. A large real-world cohort study using the TriNetX database (459,886 patients, 2022–2024) found tirzepatide was associated with a 44% higher risk of osteoporosis or fragility fracture compared to other GLP-1 receptor agonists — likely related to the degree of weight loss rather than a direct drug effect. Gastrointestinal side effects drove discontinuation rates of 16.6% in SELECT, nearly double that of placebo. And the long-term consequences of sustained GLP-1 receptor activation over decades remain genuinely unknown. These drugs are being prescribed to millions, but no trial has followed patients beyond four years.
Rapamycin and Metformin: The Longevity Drugs That Haven't Proven Themselves in Humans
The mTOR inhibitor rapamycin extends lifespan by 9–14% in mice — among the most robust results in aging biology. But a critical appraisal published in Aging in 2025 by Hands et al. concluded bluntly: no human trial has directly shown rapamycin extends life or clearly slows aging in healthy older adults. Bryan Johnson tried rapamycin as part of his protocol and discontinued it, citing elevated blood glucose, susceptibility to infection, and impaired healing — side effects well-documented at clinical doses.
The TAME trial (Targeting Aging with Metformin), led by Nir Barzilai at Albert Einstein College of Medicine, represents the field's most important regulatory gambit. Designed to enroll 3,000 subjects aged 65–79 across 14 U.S. centers, TAME would be the first trial to establish aging itself as a treatable indication at the FDA. The trial design was approved by the FDA — a significant precedent. But as of mid-2025, TAME remains only partially funded, with roughly half of its $45–70 million budget secured. Meanwhile, a 2024 study in male cynomolgus monkeys found metformin slowed aging by 4–6 years based on molecular profiles, improving cognitive function and reducing inflammation — but not through the expected mechanisms of glucose or weight control. The evidence is suggestive, but the human trial that would make it definitive keeps stalling.
The most rigorously designed animal trial currently underway is TRIAD (Test of Rapamycin In Aging Dogs), funded by a $7 million NIH NIA grant. Targeting 580 dogs across 20 sites, TRIAD is described as "the first rigorous test of a pharmacologic intervention against biological aging with lifespan and healthspan metrics as endpoints to be performed outside of the laboratory in any species." Completion is slated for 2029 — if federal budget cuts don't intervene first.
Gene Editing Enters the Clinic; Immunotherapy Reaches Solid Tumors
On December 8, 2023, the FDA approved Casgevy (exagamglogene autotemcel), the first-ever CRISPR-based gene therapy, for sickle cell disease. Developed by CRISPR Therapeutics and Vertex Pharmaceuticals, Casgevy uses CRISPR/Cas9 to edit patients' hematopoietic stem cells, inducing fetal hemoglobin expression. Among 31 patients with adequate follow-up, 93.5% achieved freedom from vaso-occlusive crises for at least 12 consecutive months. The therapy costs approximately $2.2 million per patient. Jennifer Doudna called it "a truly remarkable achievement," noting the 11-year journey from laboratory discovery to approved medicine.
Base editing and prime editing are advancing rapidly behind CRISPR. Verve Therapeutics' VERVE-102, which uses single-base editing to target the PCSK9 gene for cholesterol reduction, showed dose-dependent LDL reductions of up to 59% in its highest-dose cohort. Eli Lilly acquired Verve in June 2025, signaling major pharma's confidence in the platform. Prime Medicine's PM359, the first prime editing therapy, received FDA IND clearance in April 2024 for chronic granulomatous disease. As of February 2025, approximately 250 clinical trials involving gene-editing therapeutics were underway globally.
In cancer immunotherapy, CAR-T therapy broke through into solid tumors for the first time. Three separate CAR-T products published results in glioblastoma during 2024, including a landmark Nature paper on GD2-CAR T cells for diffuse midline gliomas. Anti-CEA CAR-T achieved 57% recurrence-free survival in colorectal liver metastases. Most remarkably, the first pediatric patient ever treated with CD19 CAR-T (at Children's Hospital of Philadelphia in 2012, under Carl June's leadership) remains cancer-free after 12 years as of 2024. The FDA approved afamitresgene autoleucel in 2024 for advanced synovial sarcoma — the first genetically modified T-cell therapy approved for any solid tumor.
Psychedelics Stumble at the FDA's Door
The psychedelic therapy movement suffered a major setback in 2024. Lykos Therapeutics submitted its NDA for MDMA-assisted therapy for PTSD after Phase 3 trials showing 67–71% of participants no longer meeting PTSD diagnostic criteria. But on June 4, 2024, the FDA's Psychopharmacologic Drugs Advisory Committee voted 9–2 against effectiveness and 10–1 against benefits outweighing risks. The FDA issued a Complete Response Letter on August 9, 2024, declining approval. Concerns centered on functional unblinding (over 90% of participants correctly guessed their treatment group), data reliability issues, and ethical violations at one trial site.
Psilocybin now appears more likely to reach approval first. Compass Pathways' Phase 3 trial for treatment-resistant depression met its primary endpoint in June 2025. As Stanford's Boris Heifets noted, psilocybin is "now poised to gain approval ahead of MDMA."
Wearables Proliferate, but Clinical Evidence Lags Behind Consumer Enthusiasm
The FDA's March 2024 clearance of the Dexcom Stelo as the first over-the-counter continuous glucose monitor marked a watershed moment for consumer health tracking. Non-diabetic users accounted for 41.46% of OTC CGM revenue in 2024, and the CGM market is projected to grow from $6.28 billion in 2025 to $17.16 billion by 2031. Yet the clinical evidence for CGMs in healthy populations remains thin. Endocrinologists have warned that non-diabetic users may "unnecessarily worry about normal blood sugar shifts," potentially generating anxiety rather than insight. The FDA itself issued a safety notice emphasizing it has "not authorized, cleared, or approved any smartwatch or smart ring that is intended to measure or estimate blood glucose values on its own."
The Key Figures: Credibility Under Scrutiny
The longevity field's most prominent figures are navigating a credibility reckoning. David Sinclair resigned as president of the Academy for Health and Lifespan Research in March 2024 after his co-founded company claimed a dog supplement was "proven to reverse aging," prompting mass resignations. The Wall Street Journal published a December 2024 exposé titled "A 'Reverse Aging' Guru's Trail of Failed Businesses." In 2025, Sinclair's lab lost its NIH R01 grant amid federal funding disputes with Harvard. Peter Attia's Outlive became a bestseller in 2023, popularizing the "Medicine 3.0" framework of proactive prevention. Retro Biosciences, backed by Sam Altman's $180 million, was reportedly raising a $1 billion Series A, demonstrating AI-enhanced stem cell reprogramming — redesigning Yamanaka factors for a 50x increase in pluripotency markers — in collaboration with OpenAI.
Part II: The Generation — How Gen Z Became the First Longevity-Native Cohort
A Generation Tracking Everything
Gen Z is not merely interested in longevity — they are instrumentalizing it. Gen Z smart wearable users jumped from 17.0 million in 2022 to 21.1 million in 2024, overtaking Gen X for the first time, with adoption growing at approximately 17% annually. Among first-time smartwatch buyers in 2025, 61% cited health monitoring as their primary motivation. Smart rings — led by Oura, which commands 80% market share — saw unit volume explode by 195% year-over-year. 53% of Gen Z now report consuming dietary supplements weekly, up from 42% in 2019, the largest gain of any generation.
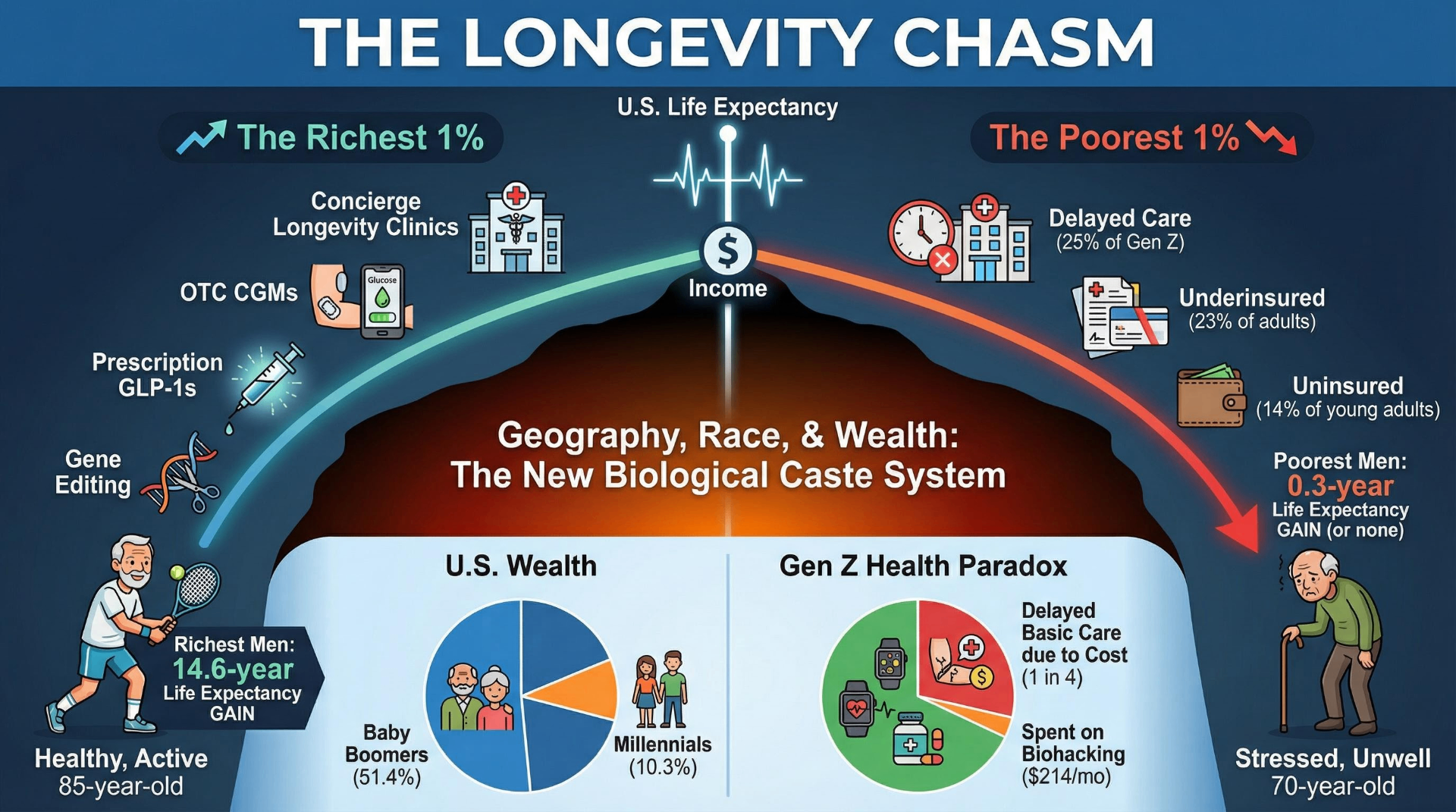
The biohacking market reflects this demand. Estimated at $24.81 billion in 2024, it is projected to reach $69.09 billion by 2030. A 2025 survey by the Sanctuary Wellness Institute found 64% of Americans tried a new biohacking method in the prior year, with top motivations split between longevity (20%), mental clarity (19%), and physical appearance (15%). Google searches for "biohacking" surged 900% over a 90-day period in 2024. The average American biohacker reported spending $214 per month on optimization — supplements, health apps, and prescription medications.
Social Media as the New Medical School
The vectors through which Gen Z receives health information have fundamentally diverged from prior generations. Social media is the number-one information source for biohacking (cited by 67% of respondents), surpassing medical studies (49%) and podcasts (44%). FitTok alone has accumulated over 64 billion views on TikTok.
Andrew Huberman's podcast became the third most popular in the U.S. on Spotify in 2023, with 7.3 million YouTube subscribers and 7.4 million Instagram followers. Bryan Johnson has transcended biohacking into cultural iconography — his Netflix documentary Don't Die: The Man Who Wants to Live Forever premiered in 2025, and his Blueprint protocol raised $60 million from investors including Naval Ravikant.
The trust problem, however, is significant. Scientists have criticized Huberman for promoting "poorly regulated" dietary supplements and making "poorly evidenced health claims." The pressure is internalized: 69% of respondents felt social media pressure to look a specific way, and 41% felt they were "falling behind" peers on wellness. The line between proactive optimization and health anxiety is razor-thin for a generation where 61% already carry an anxiety diagnosis.
The Paradox: Optimizing for 120 While Unable to Afford a Copay
The most striking tension in Gen Z's relationship to longevity is economic. 80% of Gen Z report that healthcare costs impose at least a moderate financial burden on their household — nearly twice the rate of baby boomers. One in four Gen Z adults has delayed a doctor's visit in the past three months due to cost. Only 70% report that their last healthcare visit was even partially covered by insurance, compared to 86% of boomers and seniors.
The uninsured rate among young adults aged 19–25 stands at 14.3% — the highest of any age group. Among Hispanic adults aged 18–64, nearly one in four (24.6%) lacks health insurance entirely. The juxtaposition is stark: a generation whose biohacking enthusiasts spend $214 per month on optimization exists alongside peers who skip recommended treatments because they cannot afford a cost estimate.
Prevalence data on orthorexia underscores the psychological dimension. Among fitness enthusiasts aged 18–30, a 2024 study found overall orthorexia prevalence of 53.24%, highest among gym enthusiasts (63.8%). For Gen Z, health optimization and health anxiety are not opposite ends of a spectrum — they are often the same impulse expressed through different income brackets.
Part III: The System — A Policy Infrastructure Designed for Shorter Lives
Federal Funding at a Crossroads
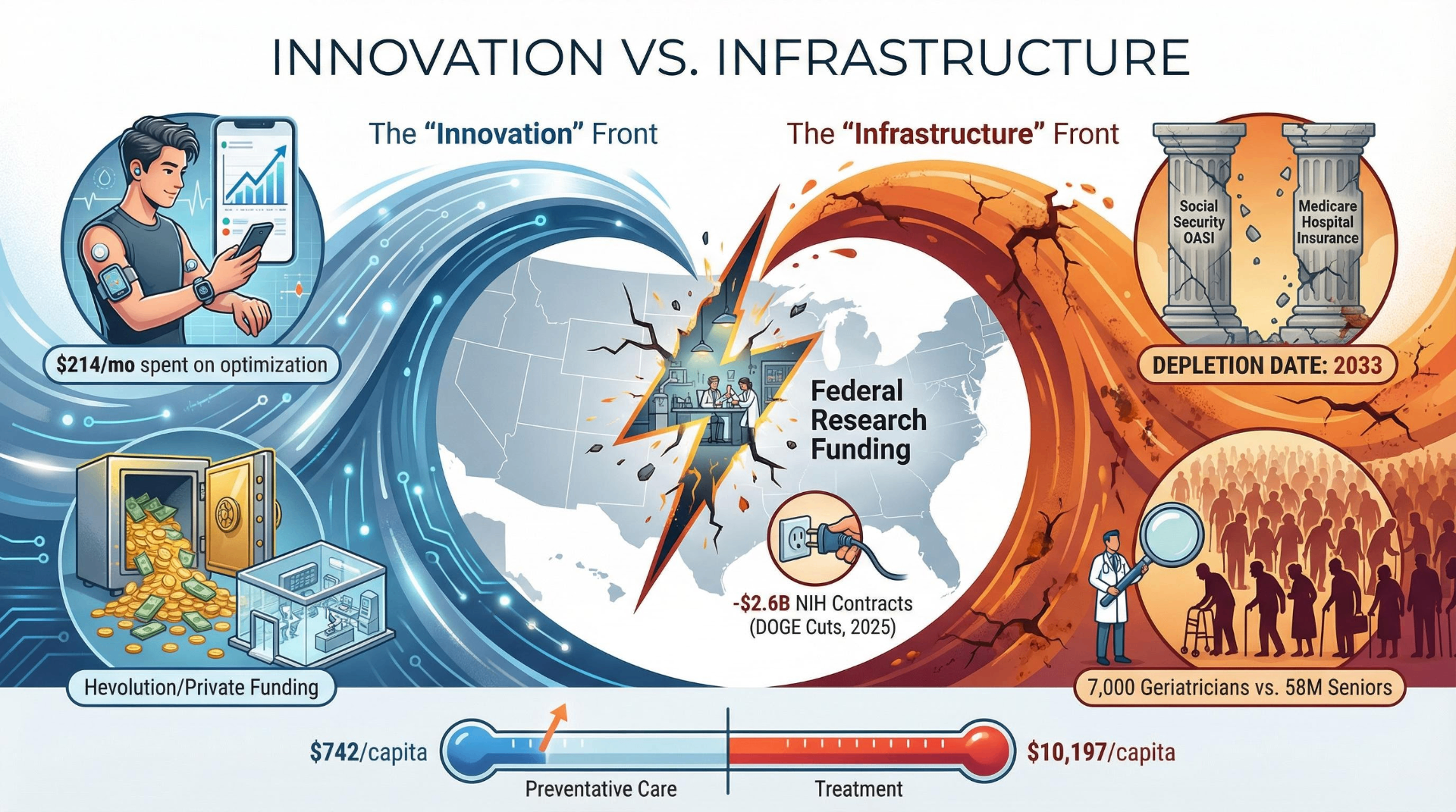
The National Institute on Aging received approximately $4.5 billion in FY2024 funding — but only about $400 million flows to basic aging biology research. The vast majority, roughly $2.6 billion, funds Alzheimer's and neuroscience-specific programs. Then came 2025. The Trump administration's DOGE ordered $2.6 billion in NIH contracts canceled in April 2025 as part of a 35% contract cut across HHS. Over 1,300 NIH employees were fired. An Implan analysis estimated these cuts could produce a $6.1 billion GDP loss and over 46,000 job losses.
The contrast with private and international investment is jarring. Private longevity companies attracted $8.49 billion in 2024. Saudi Arabia's Hevolution Foundation is authorized to spend up to $1 billion per year indefinitely on longevity research — more than double what NIA allocates to basic aging biology. CEO Mehmood Khan framed the mission: "There is not a bigger medical problem on the planet."
The FDA Still Doesn't Recognize Aging as a Disease
The single most consequential regulatory question in longevity science is whether the FDA will ever accept aging itself as a treatable indication. Currently, it does not. The TAME trial was specifically designed, with FDA input, to create such a pathway. Its composite endpoint — reduction in a basket of age-related conditions — would, if met, establish the regulatory precedent. But with the trial still only partially funded and federal science budgets under assault, the precedent remains hypothetical.
The Healthcare System Isn't Ready for the Patients It Already Has
Americans 65 and older grew from 35 million to 58 million over the past two decades and are projected to reach 85.7 million by 2050. Per-person healthcare spending for this age group was $22,356 in 2020 — 2.5 times the cost of working-age adults. The United States currently has approximately 7,000 board-certified geriatricians — roughly 0.96 per 10,000 older adults. Only one in ten U.S. medical schools requires a clinical experience in geriatrics, down from one in four in 2010.
The spending ratio tells a structural story. The U.S. spends $10,197 per capita on inpatient/outpatient care versus only $742 per capita on preventive care — a 14:1 ratio. The system is engineered to treat disease, not prevent it — precisely the opposite of what a longevity-oriented framework would require.
Twin Trust Fund Crises Converge in 2033
The 2025 Social Security Trustees Report projects the OASI trust fund will be depleted in 2033, at which point income would cover only 77% of scheduled benefits — an immediate 23% cut for every beneficiary. The 75-year unfunded obligation stands at $25.1 trillion. The worker-to-beneficiary ratio has fallen from over 5:1 in 1960 to 3:1 in 2024.
Medicare faces a parallel crisis. The 2025 Medicare Trustees Report moved the Hospital Insurance trust fund depletion date to 2033 — three years earlier than the 2024 projection. Long-term support services totaled $563.7 billion in 2023, with Medicaid absorbing $257 billion (45.6%) as the single largest payer.
The GLP-1 Coverage Battle as a Preview of Longevity Economics
Medicare spending on GLP-1 drugs soared from $57 million in 2018 to $5.7 billion in 2022 — a hundredfold increase in four years. The November 2025 GENEROUS model established negotiated prices of $245–350 per month with a $50 monthly co-pay for Medicare beneficiaries. The CBO estimates full Medicare obesity drug coverage would cost approximately $35 billion over the 2026–2034 window, partially offset by $3.4 billion in reduced medical spending. This dynamic — expensive interventions with long-term savings that the current system cannot finance short-term — will define every longevity coverage debate for decades.
The World Is Adapting Faster Than America
Japan, where 29.6% of the population is 65 or older, has incrementally restructured its entire social contract around longevity. China enacted its first retirement age reform in 70 years in September 2024. Across the OECD, employment rates for workers aged 45–64 rose by 9.3 percentage points between 2000 and 2024. The IMF concluded in 2025 that "older workers are already the main driver of GDP growth" in Europe and Japan. Thirteen OECD countries have introduced automatic links between pension eligibility and life expectancy — a mechanism the U.S. has not adopted.
Part IV: The Divide — Longevity as the New Inequality Frontier
The Gap Between Rich and Poor Is Already Measured in Years of Life
A November 2024 Lancet study updated the foundational "Eight Americas" framework into "Ten Americas," finding that life expectancy within the United States ranges from 63.6 years (American Indian/Alaska Native populations in the West) to 84.0 years (Asian Americans) — a 20.4-year gap that widened dramatically during COVID-19. Raj Chetty's landmark JAMA study documented a 14.6-year life expectancy gap for men and 10.1 years for women between the richest and poorest 1% of Americans. Between 2001 and 2014, the richest 5% gained 2.3–2.9 years of life expectancy while the poorest 5% gained 0.3 years or nothing.
The Brookings Institution found that the longevity gap is accelerating across generations: an upper-income man born in 1920 could expect to live 5 years longer than a lower-income peer; for men born in 1940, that gap doubled to 12 years. Geography compounds the disparity. Poor people in New York City live 5–6 years longer than poor people in Detroit or Gary, Indiana. Life expectancy varies by as much as 30 years between the richest and poorest U.S. counties.
The "Longevity Apartheid" Thesis Gains Scholarly Grounding
Christopher Wareham, writing in the Journal of Applied Philosophy in 2025, argued that "the benefits of longevity, if successful, would almost certainly go first to the world's best off." The Markkula Center for Applied Ethics framed the structural problem: "We live in a nation that distributes health care by money rather than need. Life extension will merely make the inequalities worse."
Yuval Noah Harari argued that "for the first time in history, economic inequality will be translated into biological inequality," creating "an upper caste of upgraded superhumans, and a massive lower class of useless people." Francis Fukuyama made similar warnings in Our Posthuman Future (2002), arguing biotechnology could undermine democratic equality through biological stratification.
The Unregulated Frontier: Young Blood, Stem Cell Tourism, and DIY Biology
Ambrosia Medical charged $8,000–12,000 for young plasma transfusions before the FDA intervened in February 2019. Bryan Johnson conducted a "multigenerational plasma exchange" with his 17-year-old son and 70-year-old father in 2023, then publicly admitted: "No benefits detected." Stem cell tourism continues to thrive despite risks — clinics in the U.S., China, India, Thailand, and Mexico market unproven treatments for conditions from Parkinson's to hair loss, charging $20,000 or more per treatment.
Intergenerational Equity and the Gerontocracy Problem
Baby Boomers own 51.4% of all U.S. wealth (Q1 2025); millennials own 10.3% despite comparable population size. In 1989, Americans under 40 held approximately 13% of household wealth; by 2020, that share had fallen to 5.9%. Median home prices increased 400% between 1990 and 2024 while median household income rose less than 200%.
If longevity technologies extend the lives of those who already hold wealth and power, the implications for generational turnover are profound. The Markkula Center warned that "people who live a radically long time may not like change... life extension, as a way of slowing or stopping generation replacement, is an intrinsically preservative and conservative activity."
The Philosophical and Religious Landscape
Leon Kass argued that finitude gives life its essential qualities — interest, seriousness, beauty, and virtue. John Harris counters that being alive is inherently good and that the desire for more life is a legitimate aspiration. Pew Research found that only 38% of Americans said they would want life extension treatments; the median "ideal lifespan" was 91 years. Religious perspectives are more diverse than commonly assumed — white evangelical Protestants were the most skeptical (62% opposing), while Black Protestants were the most open (54% viewing it favorably).
Conclusion: Four Convergences That Will Define the Next Decade
The scientific story is one of genuine but uneven progress. GLP-1 drugs have delivered hard cardiovascular outcomes data that no longevity intervention has matched. CRISPR has crossed from laboratory to clinic. But the foundational gamble — that aging itself can be treated as a condition — remains untested in humans.
The cultural story is one of a generation that has internalized the optimization imperative before the science is settled. Gen Z is the first cohort for whom longevity tracking is a consumer default. The psychological costs — orthorexia rates above 50% among fitness-oriented young adults, anxiety diagnoses at 61% — suggest that optimization culture is also a new vector for suffering.
The systemic story is one of structural unpreparedness meeting fiscal reality. Twin trust fund crises in 2033. A 14:1 ratio of treatment to prevention spending. Seven thousand geriatricians for 58 million elderly Americans. Federal research funding under assault at precisely the moment the science demands acceleration.
The ethical story is the one that binds the others together. The 14.6-year life expectancy gap between the richest and poorest Americans is not a future risk — it is the current reality. Every new longevity technology arrives into a system that distributes healthcare by money rather than need. The question is not whether life extension is possible, or even whether it is desirable, but whether we will build it as a public good or a private luxury.
The tools to extend human life are arriving. The harder question — the one this revolution has not yet answered — is whether "longer" will mean longer for everyone, or only for those who can afford it. That question is not scientific. It is political, moral, and ultimately civilizational. And it is already overdue.
Share Your Voice
Join the conversation to share your thoughts and help others understand this topic better.
Join the ConversationCommunity Feedback
No comments yet. Be the first to share your thoughts!